Customer Service and Ordering Information.Stroke: Vascular and Interventional Neurology.Journal of the American Heart Association (JAHA).Circ: Cardiovascular Quality & Outcomes.Arteriosclerosis, Thrombosis, and Vascular Biology (ATVB). 
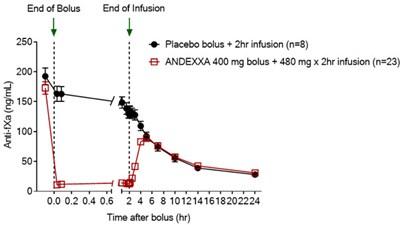
"Andexxa's rapid reversal of the anticoagulating effects of rivaroxaban and apixaban will help clinicians treat life-threatening bleeds, where every minute counts. "Today's approval represents a significant step forward in patient care and one that the medical community has been eagerly anticipating," ANNEXA-4 chair Stuart J Connolly, MD, McMaster University, Hamilton, Ontario, Canada, said in the release. The median decrease in anti-factor Xa activity from baseline was 90% for rivaroxaban and 93% for apixaban. Among the 185 evaluable high-risk patients in the open-label study, the agent was shown to provide effective clinical hemostasis in 83% of patients out to 12 hours, as recently reported by | Medscape Cardiology. Additional important safety information and full prescribing information are available here.Īpproval of the agent was supported by data from two phase 3 ANNEXA studies, ANNEXA-R and ANNEXA-A, which demonstrated a median decrease in anti-factor Xa activity from baseline of 97% for rivaroxaban and 92% for apixaban, according to a company news release, posted May 3.Īs part of its review, the FDA also assessed interim data from the ongoing ANNEXA-4 study.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
